Group 0: The Noble Gases
What are the Noble Gases?
The noble gases are a group of non-metals consisting of helium, neon, argon, krypton, xenon and radon. Radon is radioactive. The gases grow denser down the group, because the mass of the atom increases.
The elements in Group 0 are:
| Chemical Name | Chemical Symbol | Atomic Number |
| Helium | He | 2 |
| Neon | Ne | 10 |
| Argon | Ar | 18 |
| Krypton | Kr | 36 |
| Xenon | Xe | 54 |
| Radon | Rn | 86 |
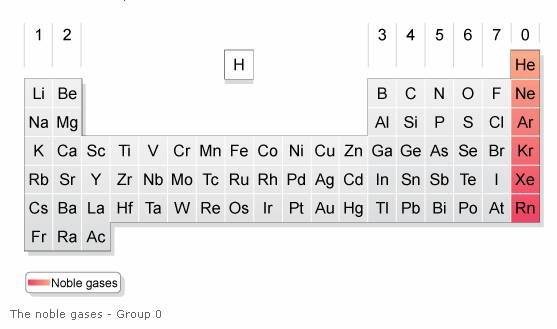
The following diagram shows the noble gases and their positions in the periodic table. Scroll down the page for more explanations about the noble gases.
What are the Properties of Noble Gases?
They are:
- Non-metals
- Colorless gases, which occur naturally in air (except for radon).
- Monatomic – they exists as single atoms
- Unreactive – they do not normally react with anything.
Why are Noble Gases unreactive?
We know that atoms react with each other to gain full outer shells of electrons. Since the atoms of noble gases have full shells already, they have no need to react. The full shells make them stable.
What are the uses of the noble gases?
The noble gases glow when a current is passed through them at low pressure. Since the noble gases are unreactive or inert, they are safe to use.
Helium is used to fill balloons and airships, because it is much lighter that air and it will not catch fire.
Argon is used to provide an inert atmosphere. For example it is used as a filler in tungsten light bulbs (we do not use air because the oxygen would make the tungsten filament burn away). We can also use it in argon lights (see the video below)
Neon is used in advertising signs. It will give red glow, but the color can be changes by mixing it with other gases.
Krypton is used in lasers and in car headlamps.
Xenon gives a bright light that looks like daylight. It is used in hospital operating theaters and car headlamps.
Helium
Helium is element number 2 on the periodic table. This video shows how helium balloons float and breathing in helium can make you sound like Donald Duck.
Neon
Neon, an inert gas, is element number 10. This video shows an example of neon light.
Argon
Argon, an inert noble gas, is element number 18. This video shows an example of argon light.
Krypton
Krypton is element number 36. The Professor explains some of the uses of Krypton.
Xenon
Xenon is the heaviest non-radioactive noble gas (5x density of air) and is used for ion thrusters, general anesthetic, and various types of lighting sources. This video shows how breathing in Xenon can lower your voice.
Radon
The Professor explains why some people are concerned about the radioactive gas, radon
Check out our most popular games!
Fraction Concoction Game:
Master fractions in the lab: mix, add, and subtract beakers to create the perfect concoction!
Fact Family Game:
Complete fact families and master the link between addition & subtraction and multiplication & division.
Number Bond Garden:
Clear the board by matching number pairs that sum to ten in this garden-themed mental math puzzle.
Online Addition Subtraction Game:
Practice your addition and subtraction skills to help the penguin find its mummy.
We welcome your feedback, comments and questions about this site or page. Please submit your feedback or enquiries via our Feedback page.



