

Identify Metal Ions
Related Topics:
More Lessons for IGCSE Chemistry
Math Worksheets
A series of free IGCSE Chemistry Activities and Experiments (Cambridge IGCSE Chemistry).
Check with your syllabus to find which tests you will need to know for the examination.
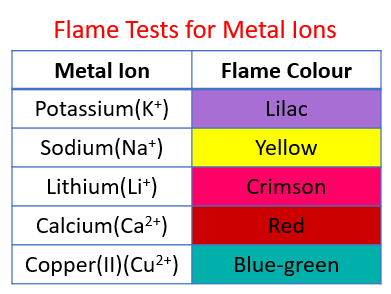
The following table shows the flame tests for metal ions:
Lithium - Crimson, Sodium - Yellow, Potassium - Lilac, Calcium - Red, Copper(II) - blue-green.
Use flame tests to identify metal ions (cations).
The following table shows the tests for aqueous cations:
Aluminium, ammonium, calcium, chromium(III), copper, iron(II), iron(III), Zinc.
Sodium hydroxide is added slowly and then in excess to solutions of these salts.
The zinc (II) and Aluminium (III) form white gelatinous hydroxide precipitates that redissolve.
Copper (II) forms a light blue hydroxide that does not redisssolve.
Iron (II) and (III), respectively, form green and brown hydroxides that do not redissolve. Test cations with aqueous Ammonia
Test Chromium(III) ions with aqueous NaOH and aqueous Ammonia


More Lessons for IGCSE Chemistry
Math Worksheets
A series of free IGCSE Chemistry Activities and Experiments (Cambridge IGCSE Chemistry).
Check with your syllabus to find which tests you will need to know for the examination.
The following table shows the flame tests for metal ions:
Lithium - Crimson, Sodium - Yellow, Potassium - Lilac, Calcium - Red, Copper(II) - blue-green.
Use flame tests to identify metal ions (cations).
Aluminium, ammonium, calcium, chromium(III), copper, iron(II), iron(III), Zinc.
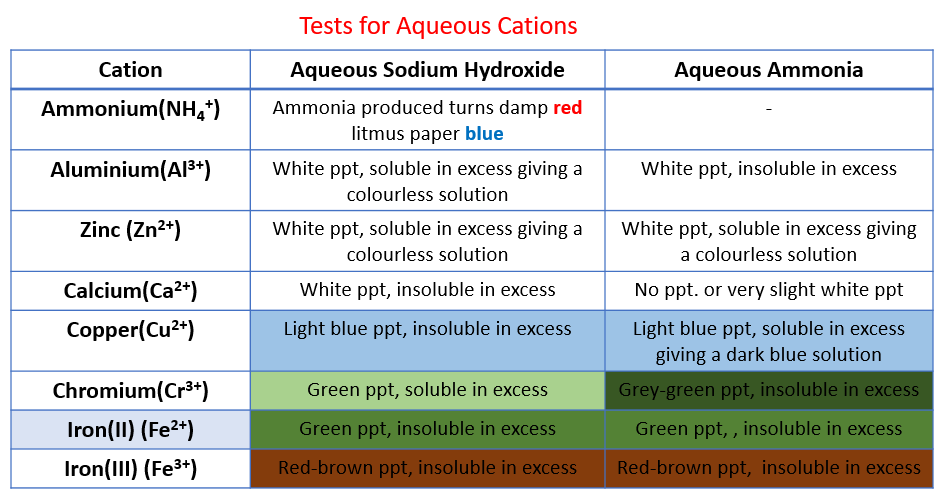
Sodium hydroxide is added slowly and then in excess to solutions of these salts.
The zinc (II) and Aluminium (III) form white gelatinous hydroxide precipitates that redissolve.
Copper (II) forms a light blue hydroxide that does not redisssolve.
Iron (II) and (III), respectively, form green and brown hydroxides that do not redissolve. Test cations with aqueous Ammonia
Test Chromium(III) ions with aqueous NaOH and aqueous Ammonia
Check out our most popular games!
Fraction Concoction Game:
Master fractions in the lab: mix, add, and subtract beakers to create the perfect concoction!
Fact Family Game:
Complete fact families and master the link between addition & subtraction and multiplication & division.
Number Bond Garden:
Clear the board by matching number pairs that sum to ten in this garden-themed mental math puzzle.
Online Addition Subtraction Game:
Practice your addition and subtraction skills to help the penguin find its mummy.


We welcome your feedback, comments and questions about this site or page. Please submit your feedback or enquiries via our Feedback page.



