

Stoichiometry Help
A series of free IGCSE Chemistry Activities and Experiments (Cambridge IGCSE Chemistry).
What is Stoichiometry?
How to use the mole and mole ratio to perform stoichiometric calculations?
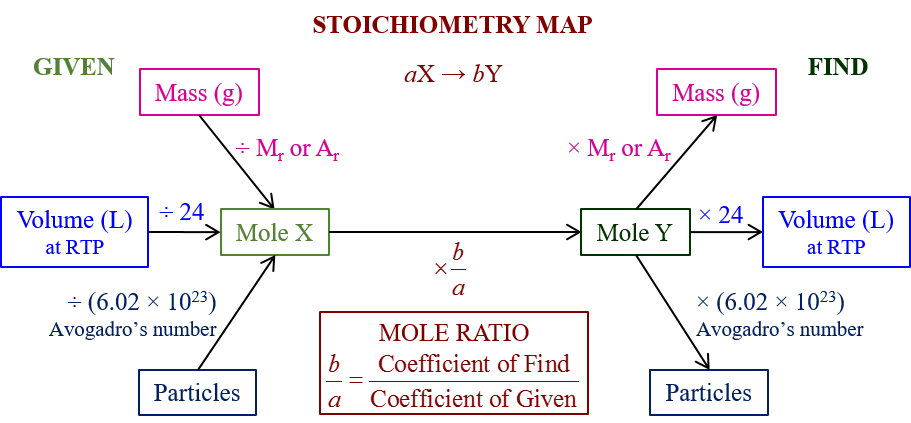
The following Stoichiometry Road Map gives a summary of how to use stoichiometry to calculate moles, masses, volumes and particles in a chemical reaction. Scroll down the page for more examples and solutions.
How to use the Stoichiometry Road Map for Gases?
The volume of one mole of any gas is 24 dm3 at room temperature and pressure (r.t.p).
(Attempt the following questions and then check your answers)
Examples:
1. What volume of oxygen is required for the complete combustion of 11.5 g of sodium?
2. What volume of carbon dioxide gas is produced from the complete thermal decomposition of 500 g of calcium carbonate?
3. What mass of calcium reacts with 72 dm3 of oxygen?
4. What volume of hydrogen gas is produced when 2 g of calcium reacts with water?
5. What volume of chlorine is needed to react with 1.12 kg of iron?
6. What volume of oxygen reacts with 510 g of ammonia?
7. What mass of potassium bromide reacts with 3 dm3 of chlorine?
8. What volume of carbon dioxide is produced when 1000 cm3 of propane (C3H8) is burnt in a large volume of oxygen? Questions:
Use the above Stoichiometry Road Map to help you answer the following questions:
1. For the reaction: 6 Li(s) + N2(g) → 2 Li3N(s)
Determine:
a) the mass of N2 needed to react with 0.536 moles of Li.
b) the number of moles of Li required to make 46.4 g of Li3N.
c) the mass in grams of Li3N produced from 3.65 g Li.
d) the number of moles of lithium needed to react with 7.00 grams of N2.
2. For the reaction:
SnO2(s) + 2 H2(g) → Sn(s) + 2 H2O(l)
Determine:
a) the mass of tin produced from 0.211 moles of hydrogen gas.
b) the number of moles of H2O produced from 339 grams of SnO2.
c) the mass of SnO2 required to produce 39.4 grams of tin.
d) the number of atoms of tin produced in the reaction of 3.00 grams of H2.
e) the mass of SnO2 required to produce 1.20 x 1021 molecules of water.
Answers



What is Stoichiometry?
How to use the mole and mole ratio to perform stoichiometric calculations?
The volume of one mole of any gas is 24 dm3 at room temperature and pressure (r.t.p).
(Attempt the following questions and then check your answers)
Examples:
1. What volume of oxygen is required for the complete combustion of 11.5 g of sodium?
2. What volume of carbon dioxide gas is produced from the complete thermal decomposition of 500 g of calcium carbonate?
3. What mass of calcium reacts with 72 dm3 of oxygen?
4. What volume of hydrogen gas is produced when 2 g of calcium reacts with water?
5. What volume of chlorine is needed to react with 1.12 kg of iron?
6. What volume of oxygen reacts with 510 g of ammonia?
7. What mass of potassium bromide reacts with 3 dm3 of chlorine?
8. What volume of carbon dioxide is produced when 1000 cm3 of propane (C3H8) is burnt in a large volume of oxygen? Questions:
Use the above Stoichiometry Road Map to help you answer the following questions:
1. For the reaction: 6 Li(s) + N2(g) → 2 Li3N(s)
Determine:
a) the mass of N2 needed to react with 0.536 moles of Li.
b) the number of moles of Li required to make 46.4 g of Li3N.
c) the mass in grams of Li3N produced from 3.65 g Li.
d) the number of moles of lithium needed to react with 7.00 grams of N2.
2. For the reaction:
SnO2(s) + 2 H2(g) → Sn(s) + 2 H2O(l)
Determine:
a) the mass of tin produced from 0.211 moles of hydrogen gas.
b) the number of moles of H2O produced from 339 grams of SnO2.
c) the mass of SnO2 required to produce 39.4 grams of tin.
d) the number of atoms of tin produced in the reaction of 3.00 grams of H2.
e) the mass of SnO2 required to produce 1.20 x 1021 molecules of water.
Answers
-
Show Solutions
1.a) 2.50 g
b) 4.00 mol
c) 6.10 g
d) 1.50 mol
2. a) 12.5 g
b) 4.50 mol
c) 50.0 g
d) 4.47x1023 atoms
e) 0.150 g
Try out our new and fun Fraction Concoction Game.
Add and subtract fractions to make exciting fraction concoctions following a recipe. There are four levels of difficulty: Easy, medium, hard and insane. Practice the basics of fraction addition and subtraction or challenge yourself with the insane level.



We welcome your feedback, comments and questions about this site or page. Please submit your feedback or enquiries via our Feedback page.