

Quantitative Chemistry
Related Topics:
More Lessons for IGCSE Chemistry
Math Worksheets
A series of free IGCSE Chemistry Lessons (Cambridge IGCSE Chemistry).
Chemical analysis and formulae, The mole and chemical formulae, The mole and chemical equations, Calculations involving gases, Moles and solution chemistry.
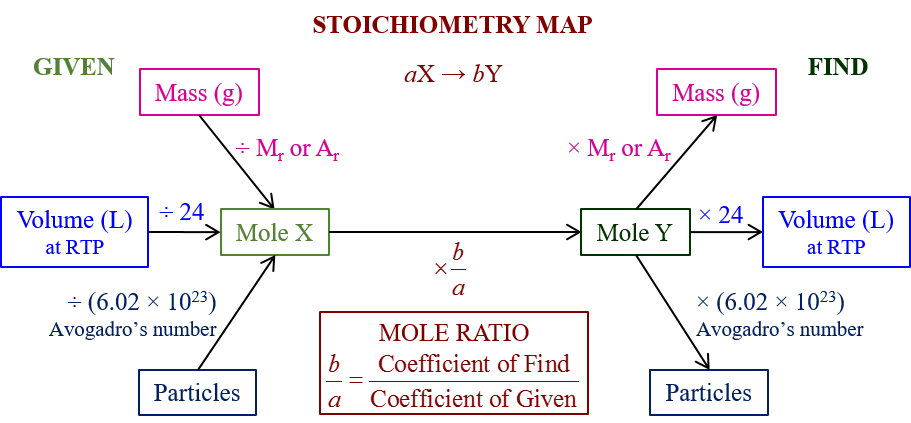
The following diagram shows how to convert between Volume, Mass, Mole and particles in a chemical equation. Scroll down the page for examples and solutions.
What is meant by the relative atomic mass for an element and how we can calculate this from isotope data. Relative Formula Mass
How to calculate relative formula mass?
It is critically important for our understanding of quantitative chemistry. Beware of a couple of pit-falls. Empirical formula from experiments
Calculating moles of an Element
What is meant by the word "mole" and how to calculate moles of an element. Calculating moles of a Compound
How to use the relative formula mass to calculate the number of moles of a compound. Using moles to balance equations
Calculating mass of a number of moles
How to calculate the mass of a given number of moles? Avogadro's constant 1
How to calculate numbers of molecules and atoms using Avogadro's constant? Avogadro's constant 2
How to calculate numbers of molecules and atoms using Avogadro's constant for a given sample? Reacting masses 1
How to use moles to calculate the masses of reactants or products in chemical reactions? Reacting masses 2
Using moles to calculate the masses of reactants or products in chemical reactions. Limiting reactant
What is a limiting reactant?
How to work out the mass of the product that we can make in a chemical reaction, even if we are not told which reactant is in excess. Calculating Percentage Yield 1
How to calculate the percentage yield for a reaction? Why percentage yield is rarely 100% and why students sometimes get this calculation wrong. Calculating Percentage Yield 2
Examples and solutions to show how to calculate the percentage yield for a reaction. Calculation involving gases
How to calculate the volume of a gas from the mass of the gas and the relative formula mass? Calculating gas volumes in chemical reactions
Concentration of Solutions
How to calculate the concentration of a solution and then the effect of changing the mass of solute and the volume of solution on the concentration Using concentration of solutions 1
How to calculate the concentration of a solution in moles per decimetre cubed? How to work out the mass of a solute dissolved in a solvent? Using concentration of solutions 2
How to calculate the concentration of solution from the volume and concentration of another solution in a chemical reaction. Titration calculations 1
How to carry out a titration calculation? Titration calculations 2
Slightly more complicated questions
Basic Calculations
Relative atomic mass, relative formula mass, percentage composition, empirical formula calculations, water of crystallisation.
Calculations involving Moles
The definition of the mole, converting masses, volumes and concentrations to moles, reacting quantity calculations, use of Avogadro's number, and calculations in electrolysis.
Molar Calculations
Determining Empirical and Molecular Formulas
How to determine the empirical and molecular formulas of a compound from quantitative analyses and includes examples of how to calculate the empirical formula given the percent composition by mass of an unknown compound


More Lessons for IGCSE Chemistry
Math Worksheets
A series of free IGCSE Chemistry Lessons (Cambridge IGCSE Chemistry).
Chemical analysis and formulae, The mole and chemical formulae, The mole and chemical equations, Calculations involving gases, Moles and solution chemistry.
The following diagram shows how to convert between Volume, Mass, Mole and particles in a chemical equation. Scroll down the page for examples and solutions.
What is meant by the relative atomic mass for an element and how we can calculate this from isotope data. Relative Formula Mass
How to calculate relative formula mass?
It is critically important for our understanding of quantitative chemistry. Beware of a couple of pit-falls. Empirical formula from experiments
Calculating moles of an Element
What is meant by the word "mole" and how to calculate moles of an element. Calculating moles of a Compound
How to use the relative formula mass to calculate the number of moles of a compound. Using moles to balance equations
How to calculate the mass of a given number of moles? Avogadro's constant 1
How to calculate numbers of molecules and atoms using Avogadro's constant? Avogadro's constant 2
How to calculate numbers of molecules and atoms using Avogadro's constant for a given sample? Reacting masses 1
How to use moles to calculate the masses of reactants or products in chemical reactions? Reacting masses 2
Using moles to calculate the masses of reactants or products in chemical reactions. Limiting reactant
What is a limiting reactant?
How to work out the mass of the product that we can make in a chemical reaction, even if we are not told which reactant is in excess. Calculating Percentage Yield 1
How to calculate the percentage yield for a reaction? Why percentage yield is rarely 100% and why students sometimes get this calculation wrong. Calculating Percentage Yield 2
Examples and solutions to show how to calculate the percentage yield for a reaction. Calculation involving gases
How to calculate the volume of a gas from the mass of the gas and the relative formula mass? Calculating gas volumes in chemical reactions
Concentration of Solutions
How to calculate the concentration of a solution and then the effect of changing the mass of solute and the volume of solution on the concentration Using concentration of solutions 1
How to calculate the concentration of a solution in moles per decimetre cubed? How to work out the mass of a solute dissolved in a solvent? Using concentration of solutions 2
How to calculate the concentration of solution from the volume and concentration of another solution in a chemical reaction. Titration calculations 1
How to carry out a titration calculation? Titration calculations 2
Slightly more complicated questions
Basic Calculations
Relative atomic mass, relative formula mass, percentage composition, empirical formula calculations, water of crystallisation.
Calculations involving Moles
The definition of the mole, converting masses, volumes and concentrations to moles, reacting quantity calculations, use of Avogadro's number, and calculations in electrolysis.
Molar Calculations
Determining Empirical and Molecular Formulas
How to determine the empirical and molecular formulas of a compound from quantitative analyses and includes examples of how to calculate the empirical formula given the percent composition by mass of an unknown compound
Check out our most popular games!
Fraction Concoction Game:
Master fractions in the lab: mix, add, and subtract beakers to create the perfect concoction!
Fact Family Game:
Complete fact families and master the link between addition & subtraction and multiplication & division.
Number Bond Garden:
Clear the board by matching number pairs that sum to ten in this garden-themed mental math puzzle.
Online Addition Subtraction Game:
Practice your addition and subtraction skills to help the penguin find its mummy.


We welcome your feedback, comments and questions about this site or page. Please submit your feedback or enquiries via our Feedback page.



