Naming Alkanes and Isomers
Related Topics:
More Lessons for IGCSE Chemistry
Math Worksheets
A series of free IGCSE Chemistry Activities and Experiments (Cambridge IGCSE Chemistry).
Naming Alkanes
Alkanes are hydrocarbons with single bonds between the carbon atoms.
The general formula is CnH(2n + 2)
Alkanes and their Isomers
Isomers are compounds with the same molecular formula but different structural formula.
Example:
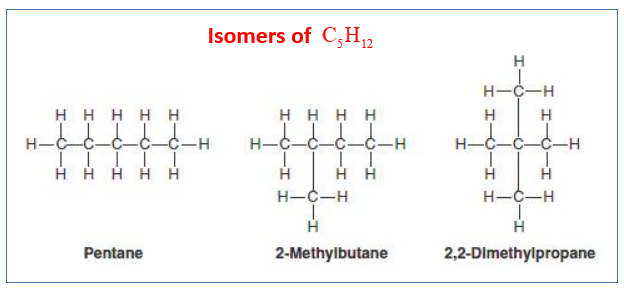
Isomers of Pentane: C5H12
n-pentane, methylbutane, dimethylpropane
Isomers of Hexane
n-hexane, 2-methypentane, 3-methypentane, 2,2-dimethybutane, 2,3-dimethybutane Questions
1. What is the key difference between the molecular and structural formula of a hydrocarbon?
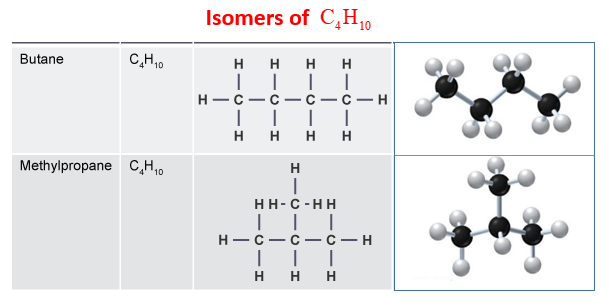
2. Name and draw the isomers of C4H10.
3. Name and draw the isomers of C5H12.
Answers


More Lessons for IGCSE Chemistry
Math Worksheets
A series of free IGCSE Chemistry Activities and Experiments (Cambridge IGCSE Chemistry).
Naming Alkanes
Alkanes are hydrocarbons with single bonds between the carbon atoms.
The general formula is CnH(2n + 2)
Isomers are compounds with the same molecular formula but different structural formula.
Example:
Isomers of Pentane: C5H12
n-pentane, methylbutane, dimethylpropane
n-hexane, 2-methypentane, 3-methypentane, 2,2-dimethybutane, 2,3-dimethybutane Questions
1. What is the key difference between the molecular and structural formula of a hydrocarbon?
2. Name and draw the isomers of C4H10.
3. Name and draw the isomers of C5H12.
Answers
-
Show Solutions for Questions 1 and 2
1. The molecular formula shows how many of each type of atom are present in a molecule. The structural formula shows the connectivity of the atoms in the molecule - the bonds and the order in which the atoms are connected.
2.
Check out our most popular games!
Fraction Concoction Game:
Master fractions in the lab: mix, add, and subtract beakers to create the perfect concoction!
Fact Family Game:
Complete fact families and master the link between addition & subtraction and multiplication & division.
Number Bond Garden:
Clear the board by matching number pairs that sum to ten in this garden-themed mental math puzzle.
Online Addition Subtraction Game:
Practice your addition and subtraction skills to help the penguin find its mummy.


We welcome your feedback, comments and questions about this site or page. Please submit your feedback or enquiries via our Feedback page.