Introduction to Atoms
Protons, Neutrons and Electrons
Atoms are incredibly small and are mostly empty space. Each atom consists of a nucleus and a cloud of particles called electrons that fly around the nucleus. The nucleus is a cluster of two kinds of particles, protons and neutrons.
All the particles in an atom are very light. Their mass is measured in atomic mass units. Protons and electrons have an electric charge, whereas neutrons do not have an electric charge.
The following table compares proton, neutron and electron in terms of their mass and charges. Scroll down the page for more examples and explanations. 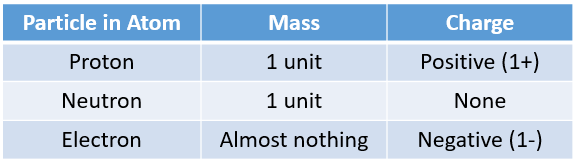
Atomic Number
The number of protons in an atom can be used to identify the atom.
Every other atom has a different number of protons. For example, since only a sodium atom has 11 protons then any atom with 11 protons would be a sodium atom.
The number of protons in an atom is called its atomic number or proton number. For example, the atomic number of sodium is 11.
Number of Electrons
Every neutral atom has an equal number of protons and electrons.
This means that the total charge on the protons cancels out the total charge on the electrons and the atom has no overall charge. For example, the sodium atom has 11 protons and 11 electrons and there is no overall charge.
Nucleon Number
Protons and neutrons, also called nucleons, are located in the nucleus of the atom. The total number of protons and neutrons in an atom is called its nucleon number. For example, the sodium atom has 11 protons and 12 neutrons. The nucleon number for the sodium atom is 11 + 12 = 23.
Isotopes
Isotopes are atoms of the same element, with different numbers of neutrons.
For example, all carbon atoms have 6 protons but some carbon atoms have more neutrons than others.
Carbon-12 has 6 protons, 6 electrons and 6 neutrons.
Carbon-13 has 6 protons, 6 electrons and 7 neutrons.
Carbon-14 has 6 protons, 6 electrons and 8 neutrons.
Carbon-12, Carbon-13 and Carbon-14 are called isotopes of carbon.
Introduction to Atoms
The atom, proton, neutron and electron
Introduction to Atomic Structure
This video shows how all atoms are made of protons, neutrons (in the nucleus) and electrons (outside the nucleus). The positive charge of the protons and the negative charge of the electrons create an attractive force that holds the atom together. The number of protons in the nucleus determines which element the atom is.
For example, all carbon atoms have 6 protons in their nuclei. Because neutral atoms carry no overall charge, the number of protons and electrons must be equal, thus canceling out their charges.
Electrons are wrapped around the nucleus in shells, with definite rules governing how many electrons each shell can hold. When atoms collide with each other, their outer shells come into contact with each other, so the number of electrons in an atom’s outer shell is incredibly important. This number of electrons determines the properties of the atom.
Check out our most popular games!
Fraction Concoction Game:
Master fractions in the lab: mix, add, and subtract beakers to create the perfect concoction!
Fact Family Game:
Complete fact families and master the link between addition & subtraction and multiplication & division.
Number Bond Garden:
Clear the board by matching number pairs that sum to ten in this garden-themed mental math puzzle.
Online Addition Subtraction Game:
Practice your addition and subtraction skills to help the penguin find its mummy.
We welcome your feedback, comments and questions about this site or page. Please submit your feedback or enquiries via our Feedback page.



